Also, you may need to indicate the state of matter (solid, liquid, aqueous, gas) of reactants and products. You may also need to balance equations for both mass and charge. This is how you balance a simple chemical equation for mass. Aqueous solutions of lead (II) nitrate and sodium chloride are mixed. 
If necessary, reduce to the lowest ratio. However, chemists always write the simplest equation, so check your work to make sure you can't reduce your coefficients. Enter the equation directly into the Balancing Chemical Equations Calculator to balance the given chemical equations. Make sure that all coefficients are in the lowest possible ratio. 
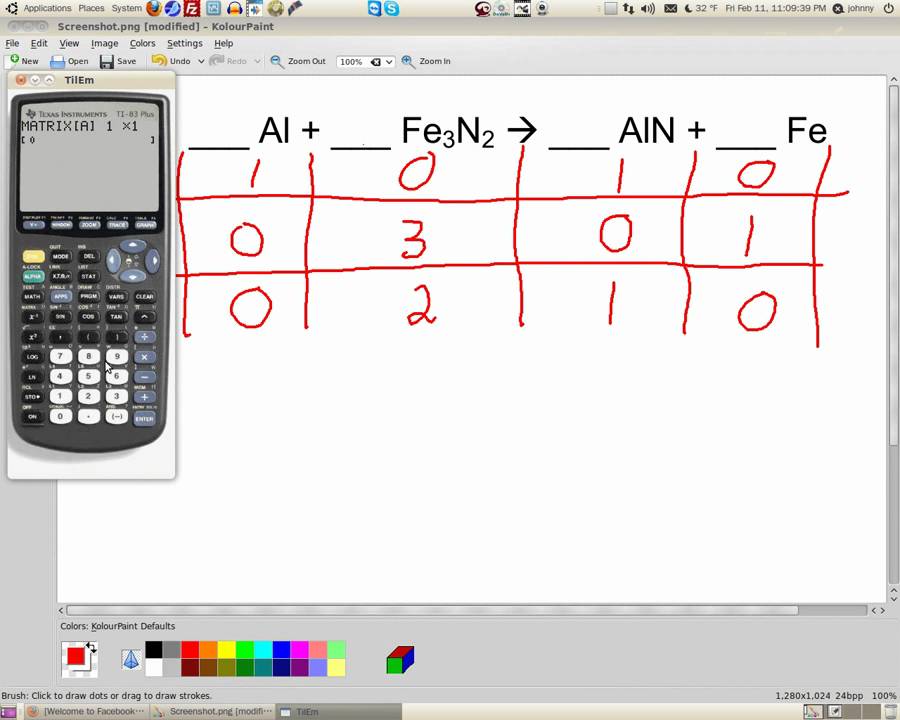
There is no one particular way to balance. Try balancing big formulas first save free elements for last. For example, if you double all of the coefficients, you still have a balanced equation: Chemical equation - Describes a chemical change. Note: You could have written a balanced equation using multiples of the coefficients. This works! The balanced chemical equation is: If you try 3 O 2, then you have 6 oxygen atoms on the reactant side and also 6 oxygen atoms on the product side. If you put a 2 in from of O 2, that will give you 4 atoms of oxygen, but you have 6 atoms of oxygen in the product (coefficient of 2 multiplied by the subscript of 3). Now, look at the equation (use inspection) to see which coefficient will work to balance oxygen. Balancing chemical equations involves the addition of stoichiometric coefficients to the reactants and products. The reason is that they usually appear in multiple reactants and products, so if you tackle them first you're usually making extra work for yourself. When balancing chemical equations, the last step is to add coefficients to oxygen and hydrogen atoms. Iron is balanced, with 4 atoms of iron on each side of the equation. BYJUS online chemical equation calculator tool makes the. By inspection (i.e., looking at it), you know you have to discard a coefficient of 2 for some higher number.ģ Fe doesn't work on the left because you can't put a coefficient in from of Fe 2O 3 that would balance it.Ĥ Fe works, if you then add a coefficient of 2 in front of the rust (iron oxide) molecule, making it 2 Fe 2O 3. balanced equation, equilibrium constant, substance properties with chemical names and formulas. While that would balance iron, you already know you're going to have to adjust oxygen, too, because it isn't balanced. There is one atom of iron on the left and two on the right, so you might think putting 2 Fe on the left would work. Iron is present in one reactant and one product, so balance its atoms first. Balance any oxygen or hydrogen atoms last.Balance atoms present in a single molecule of reactant and product first.Basically, you look at how many atoms you have on each side of the equation and add coefficients to the molecules to balance out the number of atoms. There is a strategy that will help you balance equations more quickly. As with subscripts, you don't write the coefficient of "1", so if you don't see a coefficient, it means there is one molecule. If, for example, you write 2 H 2O, that means you have 2 times the number of atoms in each water molecule, which would be 4 hydrogen atoms and 2 oxygen atoms. This happens fairly often at the end of a balancing sequence, when the placement of one coefficient balances two different elements at the same time. Placement of a two in front of the HCl balances the hydrogen and chlorine at the same time. When balancing equations, you never change subscripts. You add coefficients. Coefficients are whole number multipliers. The correctly balanced equation is: H 2 + Cl 2 -> 2HCl. Proper chemical equations should be balanced.Add Coefficients To Balance Mass in a Chemical Equation By counting the atoms of each element, we can see that the reaction is now balanced.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
